タグ: edi validator

Your Trading Partner Says You Need EDI, Now What?
You ve started a business selling products, and they re catching on. Now, a big box retailer like Walmart or Target wants to put your products on their shelves. But in order to cement that partnership, your new partner is asking you to adopt EDI. How do you get started? Defining EDI and Its Benefit...

Modern ESBs: Pros & Cons
Modern ESBs: Pros Cons As you adopt applications, including CRM, ERP, and accounting systems, you need to integrate these tools to gain a complete view of your customers and deliver the level of service they expect. An enterprise service bus (ESB) has long been one way to get all your solutions to t...

Have You Outgrown Your EDI VAN?
If you re like many companies, you have no choice but to adopt Electronic Data Interchange (EDI). Certain companies require their trading partners to use EDI, and to do business with them, you have to use EDI. One way companies get their feet wet with EDI is by turning to a Value Added Network (VAN...
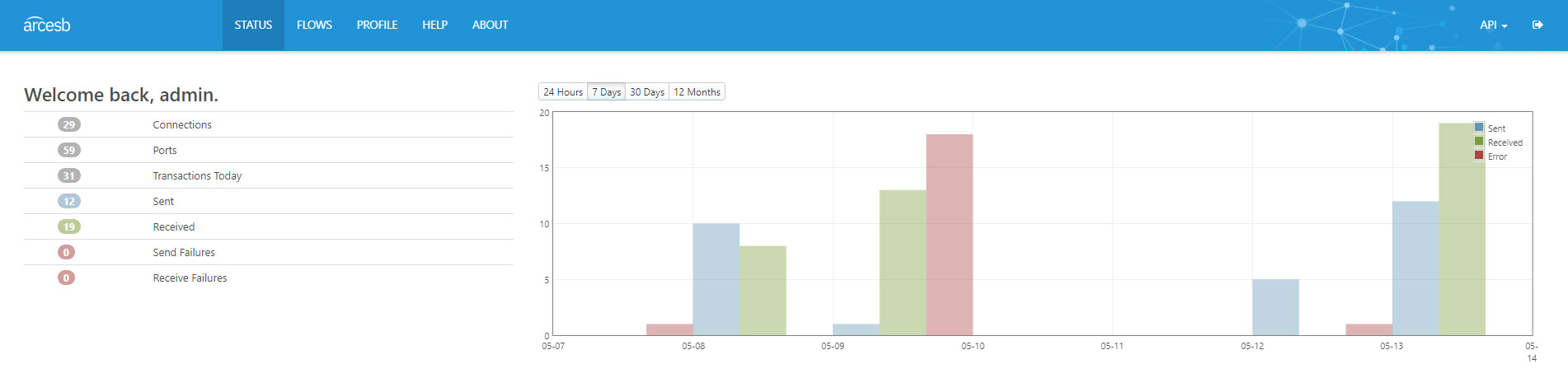
Syncrofy Partners with CData Arc for EDI Visibility
In the modern digital enterprise ecosystem, Electronic Data Interchange (EDI) has grown far beyond the simple exchange of standard business documents. The inherent complexity of vast organizations in business with thousands of vendors, suppliers, and customers across the globe means the ability to ...

First-in-First-Out: How to Fast-Track EDI Onboarding
We are often asked about the biggest challenges in onboarding EDI trading partners. While pointing out missed opportunities in a post-project review is easy, finding a strategic advantage within an ongoing concern is much, much harder. Onboarding programs are no different, historically facilitated ...
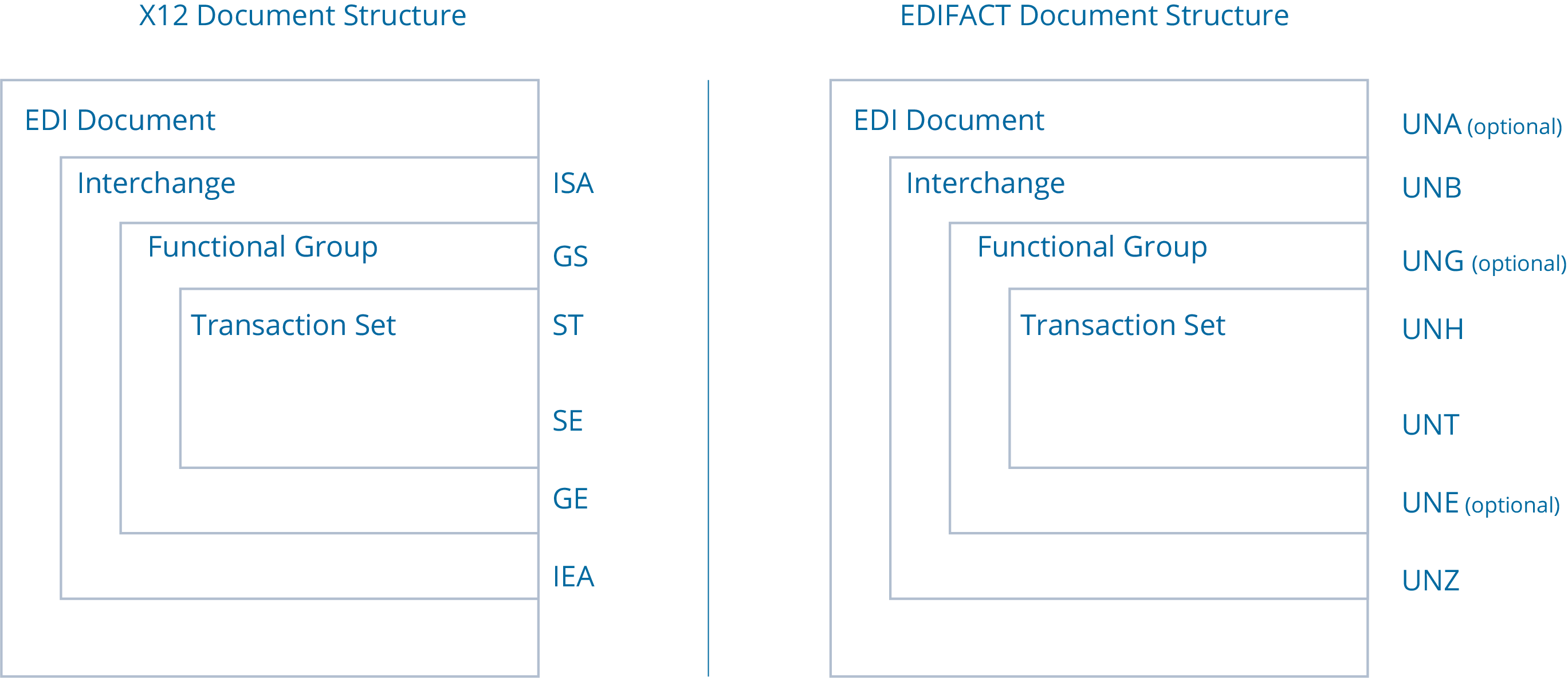
X12 vs. EDIFACT: a Complete Rundown
EDI offers many different document types. The most common are ANSI ASC X12 (X12) and UN/EDIFACT (EDIFACT). The two standards are quite similar, differing primarily in their use cases and terminology. Here are the differences and similarities between these two popular EDI document formats. Use Cases...
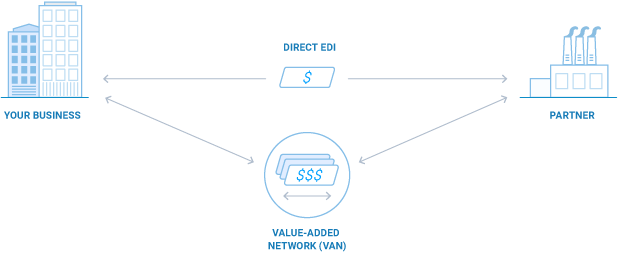
Direct EDI (AS2) vs. VANs: Pros, Cons and The Basics
Throughout the world, companies large and small alike are increasingly adding, expanding, and modernizing their EDI communications. If you need to meet partner EDI mandates or wish to capture the many benefits afforded by EDI connectivity with more of your partners, you can take several approaches ...

Walmart Adoption Nudges Blockchain EDI into the Mainstream
In September of 2019, Walmart will require more than 100 farms that supply leafy green vegetables to put detailed information about their food into a blockchain database developed by IBM. Walmart hopes to use the so-called IBM Food Trust blockchain to track this produce as it moves through the supp...
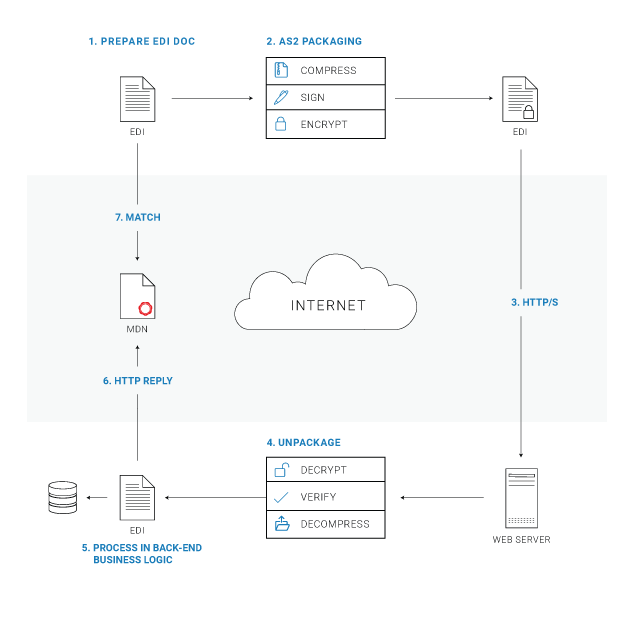
AS2 Security Basics
Applicability Statement 2, or AS2, is one of the most secure file transfer protocols for data exchange between trading partners. AS2 combines a number of secure and widely used technologies, including HTTPS, SSL Certificates, S/MIME and file hashing. How Does AS2 Security Work? Walking through the ...

CData Arc がボーイングイノベーションと中国のTitanium Powerhouse 間の商流を接続
本記事の内容は機械翻訳によるものです。記事の内容についてご質問がある場合は、 CData Arc 製品スペシャリストまでお気軽にご連絡ください 。 世界最大の原材料プロバイダーの1 つであるBaoTi Group が、CData Arc を使用してビジネスクリティカルなEDI 統合の課題をどのように解決したかをカバーする詳細なケーススタディを作成しました。BaoTi は、チタン、チタン合金、希土類金属の中国最大の生産者であり、世界最大の航空メーカーであるボーイングへの原材料の重要なサプライヤーです。BaoTi は、ボーイングの購入テクノロジーとシームレスに組み合わせて、発注書やその他のやり取...

AS4 およびAS2 通信のCData Arc 認定
本記事の内容は機械翻訳による翻訳です。記事の内容についてご質問がある場合は、 CData Arc 製品スペシャリストにご連絡ください 。 CData Arc が、AS2 とAS4 の両方のメッセージングプロトコルの2016 年第3 四半期のドラモンド認定相互運用性テストを無事に完了したことをお知らせします。CData Arc は、B2B メッセージングとMFT に最適なソリューションを提供することをお約束します。 相互運用性テストへの取り組みにより、他の認定ソリューションと接続する際のシームレスなエクスペリエンスが保証されます。 CData Arc はAS2-3Q16 ドラモンド認定相互運用...


